Temperature and Rate
-Generally, and temperature increases so does the reaction rate
-K is temperature dependent
-Generally, and temperature increases so does the reaction rate
-K is temperature dependent
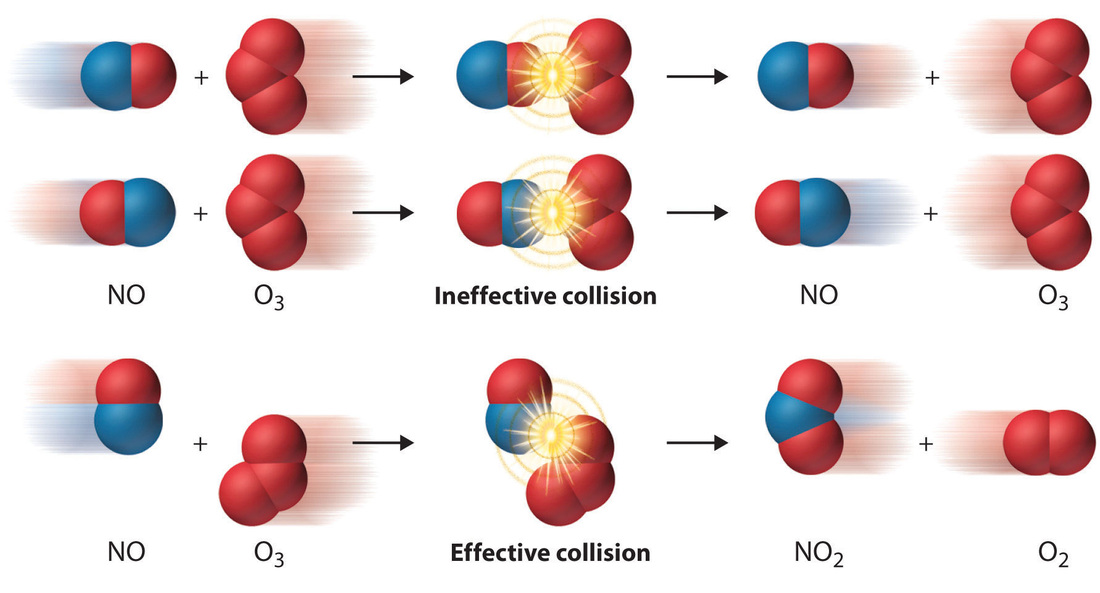
The Collision Model
-In a chemical reaction the bonds between atoms are broken and new bonds are formed
-molecules can only react if they collide with each other
-They must also collide with the correct orientation and with enough energy to break break the old bonds and form new ones.
-In a chemical reaction the bonds between atoms are broken and new bonds are formed
-molecules can only react if they collide with each other
-They must also collide with the correct orientation and with enough energy to break break the old bonds and form new ones.
Averill, Bruce, and Patricia Eldredge. Atom Collision Model. Digital image.General Chemistry: Principles, Patterns, and Applications. 2013 Flat World Knowledge, Inc., n.d. Web. 23 May 2014.
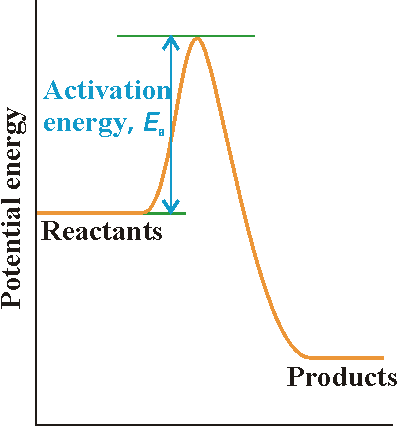
Activation Energy
-The minimum number of energy required for reaction the reaction to occur, E(a)
-The minimum number of energy required for reaction the reaction to occur, E(a)
Activation Energy Graph. Digital image. The Collision Theory and the Activated Complex Theory. N.p., n.d. Web. 23 May 2014.
-The high point of the reaction is called the transition state.
-The species present at the transition state is called the activated complex
-The energy gap between the reactants and the activated complex is the activation energy barrier.
- since the products potential energy is lower than the reactants potential energy, this reaction is exothermic.
-This means that it increases the entropy of the surroundings of the reaction.
-The enthalpy of this reaction is less than zero because it is an exothermic reaction.
-The species present at the transition state is called the activated complex
-The energy gap between the reactants and the activated complex is the activation energy barrier.
- since the products potential energy is lower than the reactants potential energy, this reaction is exothermic.
-This means that it increases the entropy of the surroundings of the reaction.
-The enthalpy of this reaction is less than zero because it is an exothermic reaction.