Catalysts
-a substance that changes the rate of reactions, but is not permanently changed itself
-a substance that changes the rate of reactions, but is not permanently changed itself
Homogeneous Catalysts
-A catalyst present in the same phase as the reacting molecules.
Heterogeneous Catalysts
-A catalyst in a different phase from the reactant molecules
-Usually a solid and the reactants gases or liquids
What do Catalysts do?
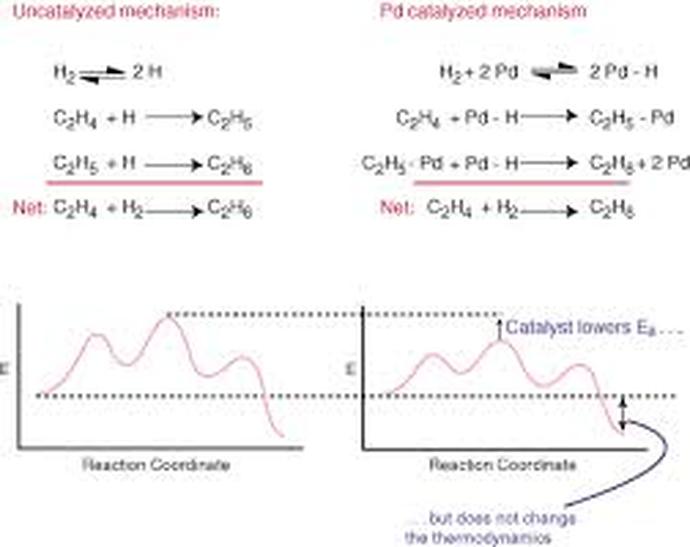
Catalysts increase the rate of a reaction by decreasing the minimum amount of energy(activation energy) the reaction needs to start the reaction.
- They change the mechanism by which the process occurs
-One way catalysts speed up a reaction is by holding the reactants together and helping bonds break
System with no Catalyst System with Catalyst
-A catalyst present in the same phase as the reacting molecules.
Heterogeneous Catalysts
-A catalyst in a different phase from the reactant molecules
-Usually a solid and the reactants gases or liquids
What do Catalysts do?
Catalysts increase the rate of a reaction by decreasing the minimum amount of energy(activation energy) the reaction needs to start the reaction.
- They change the mechanism by which the process occurs
-One way catalysts speed up a reaction is by holding the reactants together and helping bonds break
System with no Catalyst System with Catalyst
Catalyzed Mechanism Graph. Digital image. Sparknotes. SparkNotes LLC,, n.d. Web. 30 May 2014. <http://www.sparknotes.com/chemistry/kinetics/mechanisms/section1.html>
Enzymes
-enzymes are biological catalysts.
-Our body has lots of them.
Enzyme Inhibitors
-A molecule that has a strong bond with the active site of an enzyme
-blocks substrate entry and prevents reactions from happening
-enzymes are biological catalysts.
-Our body has lots of them.
Enzyme Inhibitors
-A molecule that has a strong bond with the active site of an enzyme
-blocks substrate entry and prevents reactions from happening