Determining Exponents:
If the observed reaction has:
-No change, the -Doubled, the -quadrupled, the
exponent is zero exponent is one exponent is two
If the observed reaction has:
-No change, the -Doubled, the -quadrupled, the
exponent is zero exponent is one exponent is two
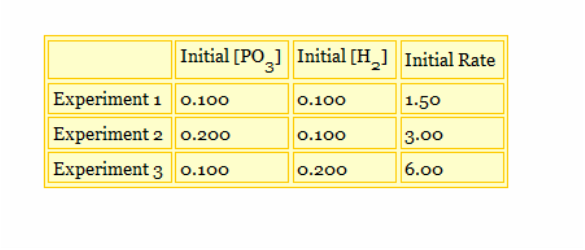
Since when the initial [PO3] is changed the reaction rate doubles. This makes its exponent 1. When you change [H2] the rate doubles. This makes that the exponent is also 1.
This means that the rate law of the reaction must be:
r=K[PO3][H2]
Rate Law Chart. Digital image. Blogspot.com. N.p., n.d. Web. 23 May 2014
This means that the rate law of the reaction must be:
r=K[PO3][H2]
Rate Law Chart. Digital image. Blogspot.com. N.p., n.d. Web. 23 May 2014
Determine the rate of reaction when [PO3]=.500M and when [H2]=.400M
You simply plug the numbers into the rate law
r=K[PO3][H2] original
r=K[.500][.400] plug in
r=.200 answer
You simply plug the numbers into the rate law
r=K[PO3][H2] original
r=K[.500][.400] plug in
r=.200 answer
